Chiral carbon in glucose12/7/2023 
As the research on these nanoparticles intensified, categorization and terminology used for these materials became a point of debate 22. In the last decade, luminescent carbon nanoparticles have appeared as a novel class of carbon-based nanomaterials. The latter area is of particular interest, since nanomaterials can provide both diagnostic and therapeutic functions combined with controllable physicochemical properties. A great interest in chiral artificial systems is attributed to a plethora of potential future applications, which range from the optical activity control of detectors/emitters 13– 15, to enantioselective catalysis 16, sensing 17, 18, bio-applications 6, 19, and medical treatment of diseases 20, 21. We refer the interested readers to several reviews devoted to the chirality in colloidal semiconductor quantum dots 5– 7, metal nanoparticles 8, nanoparticle assemblies 9, 10, liquid crystals 11, and metamaterials and metasurfaces 12. With the emergence of nanotechnology, novel pathways opened up towards the fabrication of artificial chiral objects, such as nanoparticles and their assemblies. Furthermore, plenty of bulk inorganic materials may exhibit random chirality due to asymmetry in the distribution of their defects 4. This is one of the current challenges in biophysics, which raised questions on the emergence of RNA 3. RNA has been reported to spontaneously replicate in a homochiral system, but this replication can be blocked by the presence of the opposing enantiomers. Importantly, building blocks of life 2, such as proteins, nucleic acids, glycans, and lipids are dominated by an asymmetrically distributed number of enantiomers. Many naturally occurring molecules, with amino acids being prominent examples, are chiral. 
In chemistry, chiral molecules are defined as left- or right-handed enantiomers forming sets of stereoisomers. The terms chiral and chirality originally stem from geometry and were defined by Lord Kelvin as “ I call any geometrical figure, or group of points, chiral, and say that it has chirality if its image in a plane mirror, ideally realized, cannot be brought to coincide with itself.” 1 In this terminology, chirality refers purely to the object’s geometry and does not necessarily infer any information on the physical and chemical properties of a chiral material. We then consider emerging applications of chiral carbon dots in sensing, bioimaging, and catalysis, and conclude this review with a summary and future challenges. In the main part of this review we focus on chiral carbon dots, introducing their fabrication techniques such as bottom-up and top-down chemical syntheses, their morphology, and optical/chiroptical properties. Then approaches used to induce chirality in nanomaterials are reviewed. We start this review by introducing examples of molecular chirality and its origins and providing a summary of chiroptical spectroscopy used for its characterization. In recent years, synthetic efforts leading to chiral carbon dots with other attractive optical properties such as two-photon absorption and circularly polarized light emission have flourished. Once in the blood, insulin helps glucose get to our cells.Carbon dots are luminescent carbonaceous nanoparticles that can be endowed with chiral properties, making them particularly interesting for biomedical applications due to their low cytotoxicity and facile synthesis. From there, it passes into your bloodstream. It goes into our intestines where it's absorbed. During that process, glucose is released. There, acids and enzymes break it down into tiny pieces. As we eat, food travels down our oesophagus to our stomach. It mainly comes from foods rich in carbohydrates, like bread, potatoes, and fruit. As it travels through our bloodstream to our cells, it's called blood glucose or blood sugar. Note: We should note that glucose is a type of sugar we get from foods we eat, and our body uses it for energy. There are 16 possible stereoisomers of glucose, making C as the correct option. So, from the discussion above we came to know that our correct answer is 16. Glucose has four chiral carbons in its aldehyde form, and so there are 24, or 16 possible stereoisomers of this formula. Sucrose, with nine chiral carbons, has 29 stereoisomers, or 512. For an arbitrary number (n) of chiral centers in a molecule there are as many as 2n possible stereoisomers. With 1 chiral center, there are 2 isomers, 2 chiral centers, 4 possible isomers, 3 centers, 8 isomers and 4 centers, 16 possible stereoisomers. We should know that, each time we add a chiral center to a molecule, we double the possible number of stereoisomers. 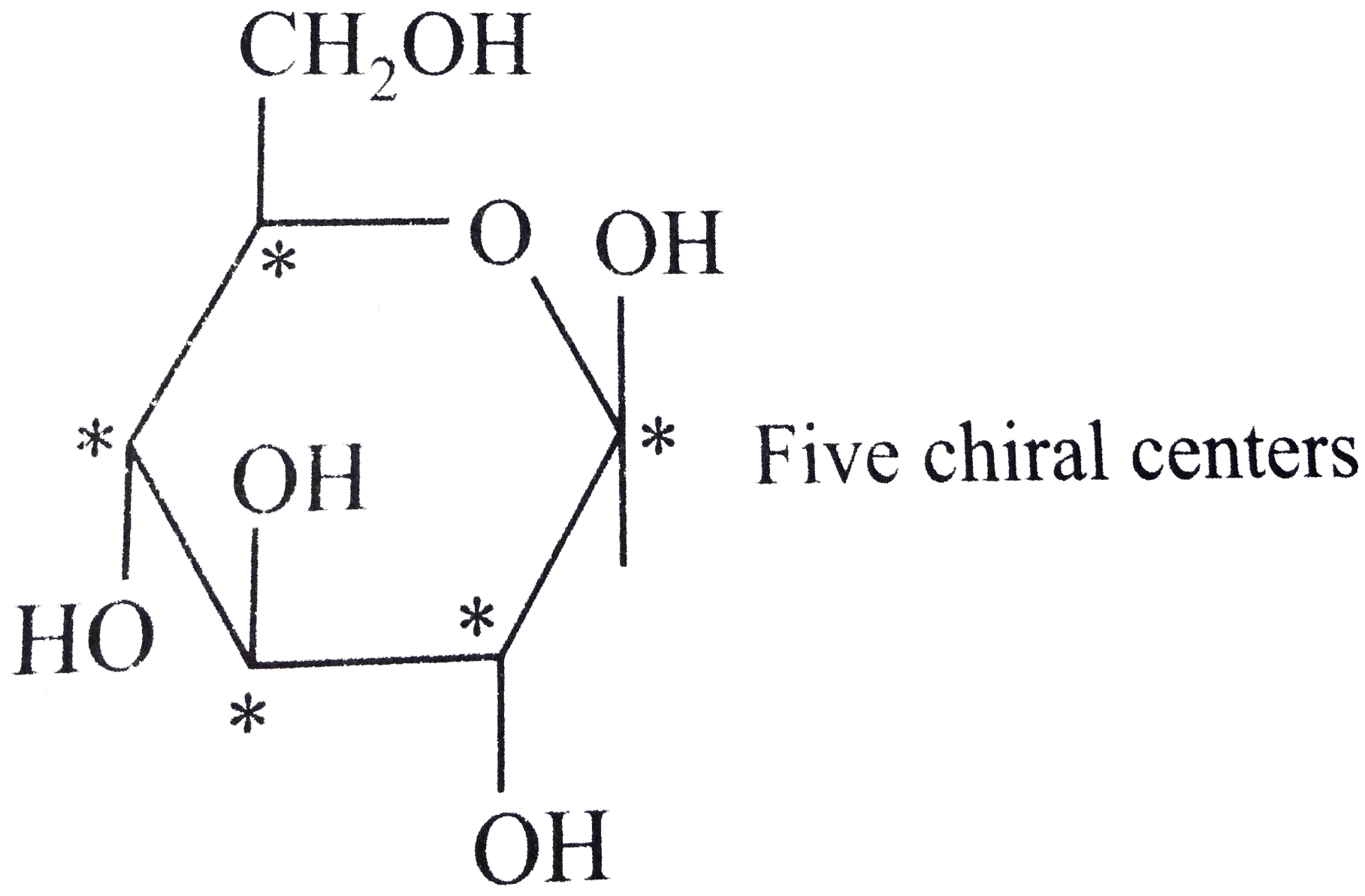
We should know that glucose is a simple sugar with the molecular formula \ Above represented structure is of glucose.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
